A) Two reactions can have identical values for DH° but very different Ea values.
B) The larger the activation energy,the slower the reaction.
C) DH° determines the height of the energy barrier.
D) The lower the activation energy,the faster the reaction.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following letters represents DH° for the forward reaction in the following energy diagram? 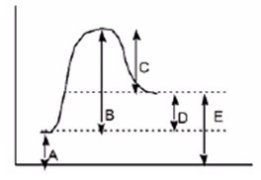
A) A
B) B
C) C
D) D
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is true?
A) Fast reactions have small rate constants.
B) Slow reactions have large rate constants.
C) A rate equation contains concentration terms for all reactants involved in a one-step mechanism.
D) A rate equation contains concentration terms for all the reactants involved in a multi-step reaction.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about addition reactions is true?
A) Two π bonds are formed.
B) Two π bonds are broken.
C) Two σ bonds are formed.
D) One π bond is formed.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about a catalyst is true?
A) A catalyst accelerates a reaction by changing the amount of reactant and product at equilibrium.
B) A catalyst accelerates a reaction by lowering the energy of activation.
C) A catalyst accelerates a reaction by raising the energy of activation.
D) A catalyst accelerates a reaction by lowering the equilibrium constant.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which reaction is fast and has Keq = 1? 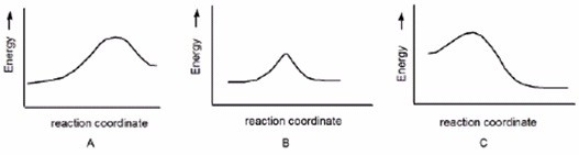
A) A
B) B
C) C
E) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the Keq corresponds to the most negative value of DG°?
A) Keq = 1
B) Keq = 101
C) Keq = 102
D) Keq = 103
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about enzymes is true?
A) Enzymes increase the activation energy for a reaction.
B) Enzymes decrease the equilibrium constant.
C) Enzymes shift the equilibrium to favor the product.
D) Enzymes lower the transition state for the rate-determining step.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about bond breaking is true?
A) Homolysis and heterolysis require energy.
B) In homolysis,the electrons in the bond are divided unequally.
C) In heterolysis,the electrons in the bond are divided equally.
D) Homolysis generates charged intermediates.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using the bond dissociation energies given,calculate DH° for the following reaction. 
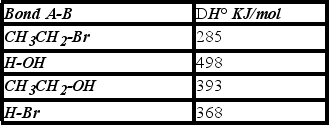
A) +108 KJ/mol
B) -130 KJ/mol
C) -22 KJ/mol
D) +22 KJ/mol
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The conversion of acetyl chloride to methyl acetate occurs via the following two-step mechanism: ![The conversion of acetyl chloride to methyl acetate occurs via the following two-step mechanism: What is the rate equation for this reaction if the first step is rate determining? A) Rate = k [acetyl chloride] [<sup>-</sup>OCH<sub>3</sub>] B) Rate = k [acetyl chloride] C) Rate = k [<sup>-</sup>OCH<sub>3</sub>] D) Rate = k [acetyl chloride] [<sup>-</sup>OCH<sub>3</sub>]<sup>2</sup>](https://d2lvgg3v3hfg70.cloudfront.net/TB7814/11eac680_9c17_cb9f_a448_7b5063864f4a_TB7814_00.jpg) What is the rate equation for this reaction if the first step is rate determining?
What is the rate equation for this reaction if the first step is rate determining?
A) Rate = k [acetyl chloride] [-OCH3]
B) Rate = k [acetyl chloride]
C) Rate = k [-OCH3]
D) Rate = k [acetyl chloride] [-OCH3]2
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which reaction is Keq > 1? 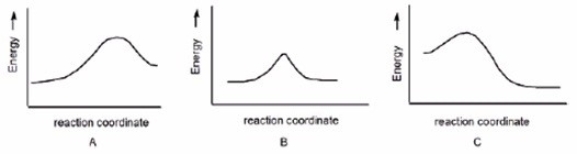
A) A
B) B
C) C
E) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If the conversion of A to B is slow and B to C is fast,what is the rate equation for this reaction? ![If the conversion of A to B is slow and B to C is fast,what is the rate equation for this reaction? A) Rate = k[(CH<sub>3</sub>) <sub>2</sub>CHCl][H<sub>2</sub>O] B) Rate = k[(CH<sub>3</sub>) <sub>2</sub>CHCl] C) Rate = k[(CH<sub>3</sub>) <sub>2</sub>CH]<sup>+</sup>[H<sub>2</sub>O] D) Rate = k[(CH<sub>3</sub>) <sub>2</sub>CH]<sup>+</sup>](https://d2lvgg3v3hfg70.cloudfront.net/TB7814/11eac680_9c17_7d7c_a448_a35f1e83c0c2_TB7814_00.jpg)
A) Rate = k[(CH3) 2CHCl][H2O]
B) Rate = k[(CH3) 2CHCl]
C) Rate = k[(CH3) 2CH]+[H2O]
D) Rate = k[(CH3) 2CH]+
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name given to the reaction species that lies at an energy minimum between steps on a reaction energy diagram?
A) Transition state
B) Activation energy
C) Reactive intermediate
D) Equilibrium product
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is not true?
A) Bond breaking is endothermic.
B) The bond dissociation energy for bond breaking is always negative.
C) Bond making is exothermic.
D) The bond dissociation energy for bond formation is always negative.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following expressions summarizes the correct relationship between the free energy change,DG°,and the equilibrium constant,Keq?
A) Keq > 1 when DG° > 0
B) Keq > 1 when DG° < 0
C) Keq < 1 when DG° < 0
D) Keq < 1 when DG° = 0
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The conversion of acetyl chloride to methyl acetate occurs via the following two-step mechanism: 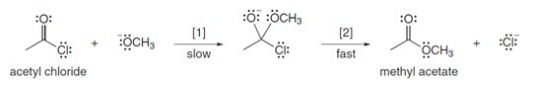 If the concentrations of both -OCH3 and acetyl chloride were increased 2 times,what would happen to the rate of the reaction?
If the concentrations of both -OCH3 and acetyl chloride were increased 2 times,what would happen to the rate of the reaction?
A) Rate would become one-fourth
B) Rate would increase 4 times
C) Rate would increase 16 times
D) Rate would increase 2 times
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The conversion of acetyl chloride to methyl acetate occurs via the following two-step mechanism: 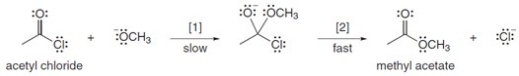 The conversion of acetyl chloride to methyl acetate would be classified as which of the following?
The conversion of acetyl chloride to methyl acetate would be classified as which of the following?
A) Addition
B) Elimination
C) Substitution
D) None of these
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The equilibrium constant for the conversion of A to D is predicted to be which of the following? 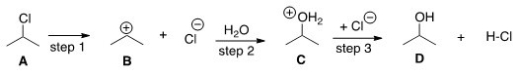
A) Keq = 1
B) Keq < 1
C) Keq > 1
D) Cannot be determined from the information provided
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which reaction is slowest? 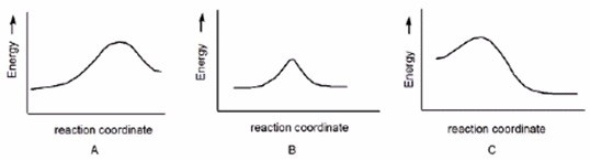
A) A
B) B
C) C
E) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 53
Related Exams