A) 286 K
B) 305 K
C) 315 K
D) 347 K
E) 3170 K
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following should have the greatest molar entropy at 298 K?
A) CH4(g)
B) H2O(l)
C) NaCl(s)
D) N2O4(g)
E) H2(g)
G) None of the above
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Which relationship or statement best describes ΔS° for the following reaction? CaO(s) + CO2(g) → CaCO3(s)
A) ΔS° ≈ 0
B) ΔS° < 0
C) ΔS° > 0
D) ΔS° = ΔH°/T
E) More information is needed to make a reasonable prediction.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which relationship or statement best describes ΔS° for the following reaction? 2NH3(g) + 2ClF3(g) → 6HF(g) + N2(g) + Cl2(g)
A) ΔS° ≈ 0
B) ΔS° < 0
C) ΔS° > 0
D) ΔS° = ΔH°/T
E) More information is needed to make a reasonable prediction.
G) C) and D)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
For a chemical reaction to be spontaneous only at high temperatures, which of the following conditions must be met?
A) ΔS° > 0, ΔH° > 0
B) ΔS° > 0, ΔH° < 0
C) ΔS° < 0, ΔH° < 0
D) ΔS° < 0, ΔH° > 0
E) ΔG° > 0
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given: H2O(l) → H2O(s) ΔH° = -6.02 kJ at 273K Calculate the entropy change of the surroundings (ΔSsurr) when one mole of water freezes at 0°C and a pressure of one atmosphere.
A) 22.1 J/K
B) -22.1 J/K
C) 397 J/K
D) -397 J/K
E) 0.022 J/K
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Elemental boron can be formed by reaction of boron trichloride with hydrogen. BCl3(g) + 1.5H2(g) → B(s) + 3HCl(g) Substance: BCl3(g) H2(g) B(s) HCl(g) S°(J/K·mol) : ? 130.6 5.87 186.8 If ΔS° = 80.3 J/K for the reaction above, what is S° for BCl3(g) ?
A) -18.2 J/K·mol
B) 18.2 J/K·mol
C) 290.1 J/K·mol
D) 355.4 J/K.mol
E) 450.6 J/K·mol
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sulfuryl dichloride is formed when sulfur dioxide reacts with chlorine. The data refer to 298 K. SO2(g) + Cl2(g) → SO2Cl2(g) Substance: SO2(g) Cl2(g) SO2Cl2(g) ΔH°f (kJ/mol) : -296.8 0 -364.0 ΔG°f (kJ/mol) : -300.1 0 -320.0 S°(J/K·mol) : 248.2 223.0 311.9 What is the value of ΔG° for this reaction at 600 K?
A) -162.8 kJ
B) -40.1 kJ
C) -28.4 kJ
D) 28.4 kJ
E) 162.8 kJ
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which relationship or statement best describes ΔS° for the following reaction? Pb(s) + Cl2(g) → PbCl2(s)
A) ΔS° ≈ 0
B) ΔS° < 0
C) ΔS° > 0
D) ΔS° = ΔH°/T
E) More information is needed to make a reasonable prediction.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction of methane with water to form carbon dioxide and hydrogen is nonspontaneous at 298 K. At what temperature will this system make the transition from nonspontaneous to spontaneous? The data refer to 298 K. CH4(g) + 2H2O(g)  CO2(g) + 4H2(g)
CO2(g) + 4H2(g)
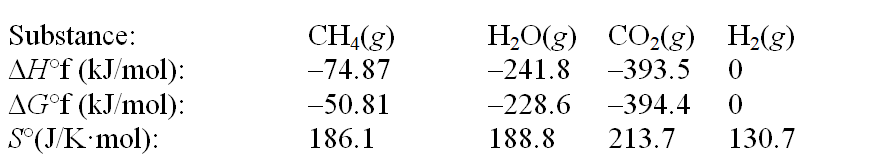
A) 658 K
B) 683 K
C) 955 K
D) 1047 K
E) 1229 K
G) C) and D)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
You are given pure samples of ammonia, NH3(g) , and nitrogen trifluoride, NF3(g) . What prediction would you make concerning their standard molar entropies at 298 K?
A) S°ammonia > S°nitrogen trifluoride
B) S°ammonia < S°nitrogen trifluoride
C) S°ammonia ≈ S°nitrogen trifluoride
D) Other conditions need to be specified before a reliable prediction can be made.
E) Even if more conditions are specified, a reliable prediction cannot be made.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In order for a process to be spontaneous,
A) ΔH must be less than zero.
B) ΔS must be greater than zero.
C) ΔG must be greater than zero.
D) it should be rapid.
E) ΔSsys + ΔSsurr must be greater than zero.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which relationship or statement best describes ΔS° for the following reaction? BaCl2(aq) + Na2SO4(aq) → BaSO4(s) + 2NaCl(aq)
A) ΔS° ≈ 0
B) ΔS° < 0
C) ΔS° > 0
D) ΔS° = H°/T
E) More information is needed to make a reasonable prediction.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A reaction has ΔG = 10.0 kJ and ΔG° = 15.0 kJ at a temperature of 50°C. Calculate the value of the reaction quotient Q under these conditions.
A) 0.16
B) 9.1 × 10-5
C) 1.1 × 104
D) 6.4
E) 6.0 × 10-6
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate ΔG° for the combustion of propane. C3H8(g) + 5O2(g) → 3CO2(g) + 4H2O(g) Substance: C3H8(g) O2(g) CO2(g) H2O(g) ΔG°f (kJ/mol) : -24.5 0 -394.4 -228.6
A) -2073.1 kJ
B) -1387.3 kJ
C) -598.5 kJ
D) 598.5 kJ
E) 2073.1 kJ
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In order for a process to be spontaneous,
A) the entropy of the system must increase.
B) the entropy of the surroundings must increase.
C) the entropy of the universe must decrease.
D) the entropy of the surroundings must decrease.
E) the entropy change of the surroundings plus the entropy change of the system must be positive.
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
As a chemical reaction proceeds toward equilibrium, the free energy of the system decreases.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which relationship or statement best describes ΔS° for the following reaction? C2H5OH(l) + 3O2(g) → 2CO2(g) + 3H2O(l)
A) ΔS° ≈ 0
B) ΔS° < 0
C) ΔS° > 0
D) ΔS° = ΔH°/T
E) More information is needed to make a reasonable prediction.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Under a given set of conditions, all microstates of a system are equally probable.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Iron(III) oxide can be reduced by carbon monoxide. Fe2O3(s) + 3CO(g)  2Fe(s) + 3CO2(g)
Use the following thermodynamic data at 298 K to determine the equilibrium constant at this temperature.
2Fe(s) + 3CO2(g)
Use the following thermodynamic data at 298 K to determine the equilibrium constant at this temperature.
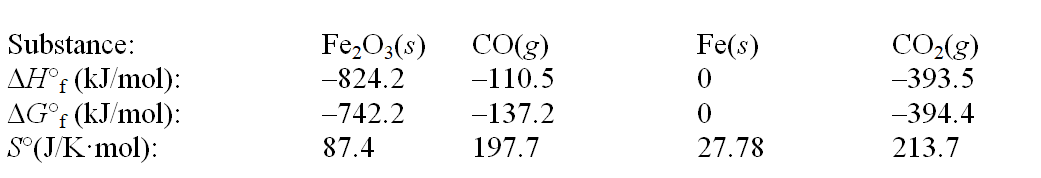
A) 7.0 × 10-6
B) 1.3 × 10-3
C) 2.2 × 104
D) 1.4 × 105
E) > 2.0 × 105
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 84
Related Exams