A) A
B) B
C) C
D) D
E) E
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When a weak acid is titrated with a strong base, the pH at the equivalence point
A) is greater than 7.0.
B) is equal to 7.0.
C) is less than 7.0.
D) is equal to the pKa of the acid.
E) is equal to 14.0 - pKb, where pKb is that of the base.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The pH of blood is 7.35. It is maintained in part by the buffer system composed of carbonic acid (H2CO3) and the bicarbonate (hydrogen carbonate, HCO3-) ion. What is the ratio of [bicarbonate]/[carbonic acid] at this pH? For carbonic acid, Ka1 = 4.2 × 10-7.
A) [bicarbonate]/[carbonic acid] = 0.11
B) [bicarbonate]/[carbonic acid] = 0.38
C) [bicarbonate]/[carbonic acid] = 2.65
D) [bicarbonate]/[carbonic acid] = 9.4
E) None of these choices are correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What volume of 0.200 M KOH must be added to 17.5 mL of 0.135 M H3PO4 to reach the third equivalence point?
A) 3.94 mL
B) 11.8 mL
C) 17.5 mL
D) 23.6 mL
E) 35.4 mL
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Write the ion product expression for silver sulfide, Ag2S.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When a strong acid is titrated with a strong base, the pH at the equivalence point
A) is greater than 7.0.
B) is equal to 7.0.
C) is less than 7.0, but is not 3.5.
D) is equal to the pKa of the acid.
E) is equal to 3.5.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following substances has the greatest solubility in water?
A) PbI2, Ksp = 7.9 × 10-9
B) BaF2, Ksp = 1.5 × 10-6
C) Ca(OH) 2, Ksp = 6.5 × 10-6
D) Zn(IO3) 2, Ksp = 3.9 × 10-6
E) Ag2SO4, Ksp = 1.5 × 10-5
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A diprotic acid H2A has Ka1 = 1 × 10-4 and Ka2 = 1 × 10-8. The corresponding base A2- is titrated with aqueous HCl, both solutions being 0.1 mol L-1. Which one of the following diagrams best represents the titration curve which will be seen? 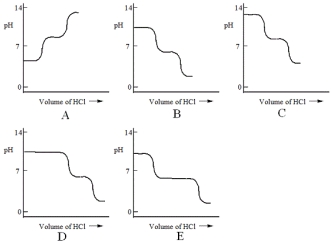
A) A
B) B
C) C
D) D
E) E
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the solubility of zinc hydroxide, Zn(OH) 2, in 1.00 M NaOH. Ksp = 3.0 × 10-16 for Zn(OH) 2, Kf = 3.0 × 1015 for Zn(OH) 42-
A) 0.60 M
B) 0.52 M
C) 0.37 M
D) 0.32 M
E) 0.24 M
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the dissolution of MnS in water (Ksp = 3.0 × 10-14) . MnS(s) + H2O(l)  Mn2+(aq) + HS-(aq) + OH-(aq)
How is the solubility of manganese(II) sulfide affected by the addition of aqueous potassium hydroxide to the system?
Mn2+(aq) + HS-(aq) + OH-(aq)
How is the solubility of manganese(II) sulfide affected by the addition of aqueous potassium hydroxide to the system?
A) The solubility will be unchanged.
B) The solubility will decrease.
C) The solubility will increase.
D) The amount of KOH added must be known before its effect can be predicted.
E) The pKa of H2S is needed before a reliable prediction can be made.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 25.0-mL sample of 0.10 M C2H3NH2 (ethylamine) is titrated with 0.15 M HCl. What is the Ph of the solution after 9.00 mL of acid have been added to the amine? Kb = 6.5 × 10-4
A) 11.08
B) 10.88
C) 10.74
D) 10.55
E) 10.49
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pH of a solution that consists of 0.50 M H2C6H6O6 (ascorbic acid) and 0.75 M NaHC6H6O6 (sodium ascorbate) ? For ascorbic acid, Ka = 6.8 × 10-5
A) 3.76
B) 3.99
C) 4.34
D) 4.57
E) 5.66
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Buffer solutions with the component concentrations shown below were prepared. Which of them should have the highest pH?
A) [H2PO4-] = 0.50 M, [HPO42-] = 0.50 M
B) [H2PO4-] = 1.0 M, [HPO42-] = 1.0 M
C) [H2PO4-] = 1.0 M, [HPO42-] = 0.50 M
D) [H2PO4-] = 0.50 M, [HPO42-] = 1.0 M
E) [H2PO4-] = 0.75 M, [HPO42-] = 1.0 M
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 25.0-mL sample of 0.35 M HCOOH is titrated with 0.20 M KOH. What is the pH of the solution after 25.0 mL of KOH has been added to the acid? Ka = 1.77 × 10-4
A) 4.00
B) 3.88
C) 3.63
D) 3.51
E) 3.47
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
If the pH of a buffer solution is greater than the pKa value of the buffer acid, the buffer will have more capacity to neutralize added base than added acid.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is the best representation of the titration curve that will be obtained in the titration of a weak base (0.10 mol L-1) with HCl of the same concentration? 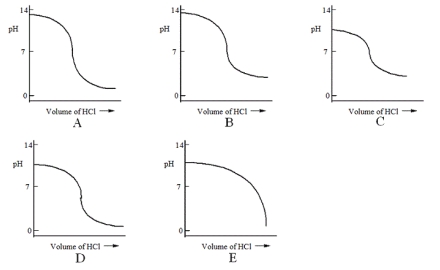
A) A
B) B
C) C
D) D
E) E
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
You need to prepare a buffer solution with a pH of 4.00, using NaF and HF. What ratio of the ratio of [base]/[acid] should be used in making the buffer? For HF, Ka = 7.2 × 10-4.
A) [base]/[acid] = 0.14
B) [base]/[acid] = 0.42
C) [base]/[acid] = 2.36
D) [base]/[acid] = 7.20
E) None of these choices are correct.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A saturated solution of calcium hydroxide, Ca(OH) 2, is in contact with excess solid Ca(OH) 2. Which of the following statements correctly describes what will happen when aqueous HCl (a strong acid) is added to this mixture, and system returns to equilibrium? (For Ca(OH) 2, Ksp = 6.5 × 10-6)
A) The solubility of Ca(OH) 2 will be unchanged.
B) The OH- concentration will decrease and the Ca2+ concentration will increase.
C) The OH- concentration will increase and the Ca2+ concentration will decrease.
D) The concentrations of both Ca2+ and OH- will increase.
E) The solubility of Ca(OH) 2 will decrease.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The solubility of calcium chromate is 1.56 × 10-3 g/100 mL of solution. What is the Ksp for CaCrO4?
A) 2.4 × 10-4
B) 1.5 × 10-5
C) 7.6 × 10-6
D) 1.0 × 10-8
E) < 1.0 × 10-8
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solution is prepared by dissolving 20.0 g of K2HPO4 and 25.0 g of KH2PO4 in enough water to produce 1.0 L of solution. What is the pH of this buffer? For phosphoric acid (H3PO4) , Ka2 = 6.2 × 10-8.
A) 7.70
B) 7.42
C) 7.21
D) 7.00
E) 6.72
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 114
Related Exams