A) trigonal bipyramidal
B) hexagonal
C) tetrahedral
D) see-saw
E) octahedral
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Boron never achieves an octet in any of its compounds.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
List all possible molecular geometries (shapes) for a nonpolar molecule with the formula AX4.
A) tetrahedral
B) seesaw
C) square planar
D) either tetrahedral or square planar
E) All of these choices are possible.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory, a molecule with the general formula AX4 will have a ______ molecular shape.
A) bent
B) trigonal planar
C) trigonal pyramidal
D) square planar
E) tetrahedral
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the following Lewis structure for phosphate, phosphorus has a formal charge of ____ and an oxidation number of ____. 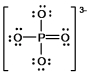
A) 0, -3
B) 0, 5
C) 5, -3
D) 5, 5
E) 3, 5
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Essay
Draw Lewis structures, showing all valence electrons, for: A) N B) Br¯ c. O2 d. SO42¯
Correct Answer

verified
Correct Answer
verified
Essay
Draw Lewis structures, showing all valence electrons, for the following species: A) S2¯ b. CO C) SO2 d. CH3OH
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Phosphoryl iodide is used in the preparation of organophosphorus derivatives and phosphate esters. Select the Lewis structure for POI3 which minimizes formal charges.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydrazine, N2H4, is a good reducing agent that has been used as a component in rocket fuels. Select its Lewis structure.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) None of these choices is correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Explain what is meant by "dipole moment", and give an example of a molecule which has polar bonds but which does not itself have a dipole moment.
Correct Answer

verified
A dipole moment arises in a molecule whe...View Answer
Show Answer
Correct Answer
verified
View Answer
True/False
In order for a non-cyclic triatomic molecule to be bent, VSEPR theory requires that there must be two lone pairs on the central atom.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory, a molecule with the general formula AX3 will have a ______ molecular shape.
A) linear
B) bent
C) trigonal planar
D) tetrahedral
E) trigonal pyramidal
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of ClCN as predicted by the VSEPR theory? (Carbon is the central atom.)
A) linear
B) bent
C) angular
D) trigonal
E) None of these choices is correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
In a Lewis structure for a molecule or ion, the sum of the formal charges on the atoms is equal to the charge on the molecule or ion.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The best Lewis structure for sulfuric acid has zero formal charges, sulfur as the central atom, and no bonds between S and H. How many single and double bonds, respectively, are there in this Lewis structure?
A) 2 single, 4 double
B) 4 single, 2 double
C) 4 single, no double
D) 6 single, no double
E) 5 single, 1 double
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many faces and how many vertexes (corners) , respectively, are there in a trigonal bipyramid?
A) 4 and 4
B) 5 and 5
C) 5 and 6
D) 6 and 5
E) 6 and 8
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Thionyl chloride is used as an oxidizing and chlorinating agent in organic chemistry. Select the best Lewis structure for SOCl2.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) None of these structures is suitable for SOCl2.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following molecules does not have a dipole moment?
A) CS2
B) H2S
C) CH2Cl2
D) PH3
E) CH2O
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Considering all the bonds in a molecule with trigonal bipyramidal geometry, what are the bond angles present?
A) 120 only
B) 90 only
C) 180 only
D) 60 and 90 only
E) 90 , 120 and 180
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of ClF4¯ as predicted by the VSEPR theory?
A) square pyramidal
B) square planar
C) see-saw
D) octahedral
E) tetrahedral
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 108
Related Exams