A) technetium-93.
B) molybdenum-89.
C) palladium-97.
D) rhenium-93.
E) ruthenium-92.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Balance the equation
A)
B)
C)
D)
E) None of the above
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A rock contains 0.37 mg of Pb-206 and 0.95 mg of U-238. The half-life of the decay series U-238 Pb-206 is 4.5 * 109 yr. Assuming no Pb-206 was present in the rock initially, how old is the rock
A) 1.7 * 109 yr
B) 5.2 * 109 yr
C) 2.7 * 106 yr
D) 4.5 * 109 yr
E) 2.4 * 109 yr
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following decay series: 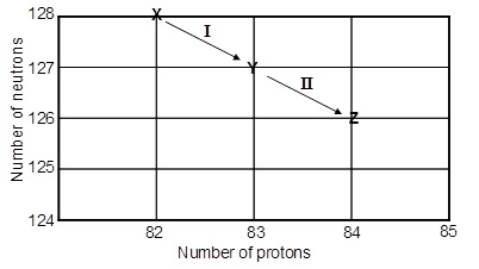 What type of nuclear process occurs at the transformation labeled I
What type of nuclear process occurs at the transformation labeled I
A) aemission
B) bemission
C) positron emission
D) electron capture
E) gamma radiation
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Balance the equation
A) Y
B) Y
C) Y
D) Y
E) None of the above
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The rates of radioactive decay processes, like the rates of chemical reactions, are sensitive to temperature changes.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Estimate the age of a bottled wine that has a tritium, 3H, content 60% that of freshly bottled wine. Tritium decays by beta decay and has a half-life of 12.3 yr.
A) 0.029 yr
B) 7.4 yr
C) 9.1 yr
D) 16 yr
E) 35 yr
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Find the nuclear binding energy of potassium-40 (atomic mass = 39.9632591 amu) in units of joules per nucleon. [Data: neutron mass = 1.674928 * 10-24 g; proton mass = 1.672623 * 10-24g; electron mass = 9.109387 * 10-28 g; NA = 6.0221367 * 1023 /mol; c = 2.99792458 * 108 m/s]
A) 1.37 * 10-12 J/nucleon
B) 5.48 * 10-11 J/nucleon
C) 5.64 * 10-11 J/nucleon
D) 1.41 * 10-12 J/nucleon
E) 2.97 * 10-12 J/nucleon
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Decay of tin-110 by electron capture yields
A) tin-109.
B) cadmium-106.
C) indium-110.
D) antimony-110.
E) tellurium-114.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
On a plot of the number of neutrons versus the number of protons, a nucleus of aluminum-28 lies above the belt of stability. During the radioactive decay of aluminum-28 a beta particle is emitted, what isotope is also formed
A)
B)
C)
D)
E) None of the above
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Compared to chemical reactions nuclear reactions, in terms of the quantity of energy released, have approximately one thousand times greater energy production for comparable amounts.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
As a result of beta decay, the product nucleus is
A) one atomic number lower than the original element.
B) two atomic numbers higher than the original element.
C) one atomic number higher than the original element.
D) two atomic numbers lower than the original element.
E) four atomic numbers lower than the original element.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
A plot of the number of neutrons versus the number of protons in various isotopes produces a "belt of stability." Isotopes below the belt of stability (i.e., with a smaller neutron-to-proton ratio) decay by beta particle emission.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete and balance the nuclear equation ________
A)
B)
C)
D)
E) None of the above
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What role does cadmium metal (Cd) play in a nuclear reactor
A) slows down the fission neutrons (moderator)
B) transfers heat from the reactor to the heat exchanger (primary coolant)
C) controls chain reaction (control rods)
D) transfers heat from the condenser to the environment (cooling tower)
E) undergoes fission (fuel rods)
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following decay series: Which type of nuclear process occurs in steps c and d, respectively
A) aemission, a emission
B) aemission, b emission
C) bemission, positron emission
D) bemission, a emission
E) positron emission, b emission
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A typical radius of an atomic nucleus is about
A) 100 µm
B) 5000 mm
C) 100 nm
D) 5 * 10-3 pm
E) 500 pm
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following decay series: 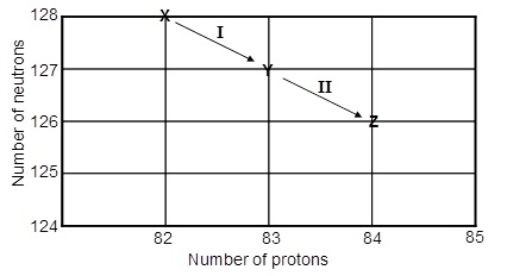 What type of nuclear process occurs at the transformation labeled II
What type of nuclear process occurs at the transformation labeled II
A) aemission
B) bemission
C) positron emission
D) electron capture
E) gamma radiation
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Rb-87/Sr-87 method of dating rocks is often used by geologists: t1/2 = 6.0 * 1010 yr.Estimate the age of a rock sample in which the present-day mole ratio of Rb-87 to Sr-87 is 36:1.
A) 2.4 * 109 yr
B) 1.7 * 109 yr
C) 3.1 * 1011 yr
D) 4.1 * 10-11 yr
E) 3.6 * 1011 yr
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The definition of free radicals is molecular fragments having one or more unpaired electrons.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 128
Related Exams