A) +1
B) −8
C) +5
D) −2
E) +3
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If aqueous solutions of lead(II) nitrate and sodium sulfate, which insoluble precipitate is formed?
A) NaNO3
B) Na2NO3
C) PbSO4
D) Pb2SO4
E) PbS
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the following equation is balanced with smallest whole number integers, what is the coefficient for HNO3? 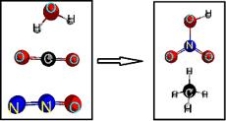
A) 1
B) 2
C) 3
D) 4
E) 5
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Once the following equation is balanced with the smallest set of whole number coefficients, what is the sum of the coefficients? (Don't forget to include coefficients of one.) __ SF4 + __ H2O → __ H2SO3 + __ HF
A) 4
B) 6
C) 7
D) 9
E) None of these answers is correct.
G) B) and D)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
What is the coefficient of H2O when the following equation is properly balanced with the smallest set of whole numbers? ___ PCl3(l) + ___ H2O(l) → ___ H3PO3(aq) + ___ HCl(aq)
A) 1
B) 2
C) 3
D) 5
E) None of these answers is correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following equations is not a redox reaction?
A) 2H2(g) + O2(g) → 2H2O(l)
B) Zn(s) + H2SO4(aq) → ZnSO4(aq) + H2(g)
C) H2O(l) + NH3(g) → NH4+(aq) + OH−(aq)
D) 6FeSO4(aq) + K2Cr2O7(aq) + 7H2SO4(aq) → Cr2(SO4) 3(aq) + 3Fe2(SO4) 3(aq) + K2SO4(aq) + 7H2O(l)
E) Cl2(g) + 2KBr(aq) → Br2(l) + 2KCl(aq)
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If aqueous solutions of Pb(NO3) 2 and NaCl are mixed, which ions, if any, are spectator ions? Pb(NO3) 2(aq) + 2NaCl(aq) → PbCl2(s) + 2NaNO3(aq)
A) Pb2+(aq) , Cl−(aq)
B) Na+(aq) , NO3− (aq)
C) Pb2+(aq) , NO3−(aq)
D) Na+(aq) , Cl−(aq)
E) None are spectator ions.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the net ionic equation for the reaction that occurs when aqueous solutions of Pb(NO3) 2 and NH4Cl are mixed?
A) Pb(NO3) 2(aq) + 2NH4Cl(aq) ? 2NH4NO3(aq) + PbCl2(s)
B) Pb2+(aq) + 2Cl?(aq) ? PbCl2(s)
C) Pb2+(aq) + 2NO3?(aq) + 2NH4+(aq) + 2Cl?(aq) ? 2NH4+(aq) + 2NO3?(aq) + PbCl2(s)
D) NH4+(aq) + NO3?(aq) ? NH4NO3(s)
E) NH4+(aq) + NO3?(aq) ? 2NO(g) + 2H2O(l)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the oxidation number of iodine in I2?
A) −1
B) 0
C) +1
D) +7
E) −7
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which chemical equation describes an acid-base neutralization reaction?
A) 2Al(s) + 3H2SO4(aq) → Al2 (SO4) 3 (aq) + 3H2(g)
B) 2SO2(g) + 2H2O(l) + O2(g) → 2H2SO4(aq)
C) LiOH(aq) + HNO3(aq) → LiNO3(aq) + H2O(l)
D) 2KBr(aq) + Cl2(g) → 2KCl(aq) + Br2(l)
E) CaBr2(aq) + H2SO4(aq) → CaSO4(s) + 2HBr(g)
G) A) and D)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Select the compound in which sulfur has its highest possible oxidation number.
A) H2S
B) SO2
C) SCl2
D) H2SO3
E) Na2SO4
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the coefficient of H2SO4 when the following equation is properly balanced with the smallest set of whole numbers? ___ Ca3(PO4) 2 + ___ H2SO4 → ___ CaSO4 + ___ H3PO4
A) 3
B) 8
C) 10
D) 11
E) None of these answers is correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which equation describes a redox reaction?
A) 2Al(s) + 3H2SO4(aq) → Al2(SO4) 3(aq) + 3H2(g)
B) 2KBr(aq) + Pb(NO3) 2(aq) → 2KNO3(aq) + PbBr2(s)
C) CaBr2(aq) + H2SO4(aq) → CaSO4(s) + 2HBr(g)
D) H+(aq) + OH−(aq) → H2O(l)
E) CO32−(aq) + HSO4−(aq) → HCO3−(aq) + SO42−(aq)
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If aqueous solutions of KMnO4, FeSO4 and H2SO4, are mixed, which ions are spectator ions? 2KMnO4(aq) + 10FeSO4(aq) + 8H2SO4(aq) → K2SO4(aq) + 2MnSO4(aq) + 5Fe2(SO4) 3(aq) + 8H2O(l)
A) only K+
B) only SO42−
C) only K+ and SO42−
D) only K+, SO42−, and Fe2+
E) only K+, SO42−, Fe2+, and Mn2+
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which salt is produced by the neutralization of hydrobromic acid with magnesium hydroxide?
A) MgBr
B) Mg2Br3
C) MgBr2
D) Mg3Br2
E) Mg2Br
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the oxidation number of N in N2H4?
A) +4
B) +2
C) −2
D) −4
E) 0
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which balanced chemical equation could represent the following result of a precipitation reaction? 
A) 2K3PO4(aq) + 3Cr2(SO4) 3(aq) → CrPO4(s) + 6KCl(aq)
B) MgSO4(aq) + 2AgNO3(aq) → Ag2SO4(s) + Mg(NO3) 2(aq)
C) MgCl2(aq) + 2AgC2H3O2(aq) → 2AgCl(s) + Mg(C2H3O2) 2(aq)
D) K2CrO4(aq) + 2AgNO3(aq) → Ag2CrO4(s) + 2KNO3(aq)
E) K2S(aq) + Pb(NO3) 2(aq) → PbS(s) + 2KNO3(aq)
G) C) and E)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Which is the net ionic equation for the reaction between aqueous solutions of lithium hydroxide and hydrobromic acid? LiOH(aq) + HBr(aq) → H2O(l) + LiBr(aq)
A) LiOH(aq) → Li+(aq) + OH−(aq)
B) HBr(aq) → H+(aq) + Br−(aq)
C) H+(aq) + OH−(aq) → H2O(l)
D) Li+(aq) + Br−(aq) → LiBr(aq)
E) Li+(aq) + OH−(aq) + H+(aq) + Br−(aq) → H2O(l) + LiBr(aq)
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If aqueous solutions of Na2CO3 and BaCl2 are mixed, which insoluble precipitate is formed?
A) Ba2CO3
B) BaCO3
C) NaCl
D) NaCl2
E) BaO
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is oxidized in the following reaction? Fe + Ag2O → FeO + 2Ag
A) Ag
B) Ag2O
C) Fe
D) FeO
E) 2Ag
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 65
Related Exams