A) 138 pm
B) 1.95 * 10-8 nm
C) 1.95 * 10-8 cm
D) 154 pm
E) 0.109 nm
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
How much enthalpy is necessary to heat 10.0 g of solid benzene (C6H6)at 0.0°C to benzene vapor at 100°C? 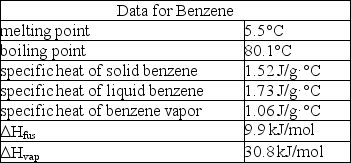
Correct Answer

verified
Correct Answer
verified
True/False
The freezing point of a liquid does not change as the atmospheric pressure changes.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
Osmium tetroxide, OsO4, is a soft crystal that melts at 40°C. The liquid does not conduct electricity. What kind of crystal is this?
Correct Answer

verified
Correct Answer
verified
Short Answer
Of the pair of compounds given, which would have the stronger intermolecular forces of attraction? H2S or H2Se
Correct Answer

verified
Correct Answer
verified
Short Answer
Identify the dominant (strongest)type of intermolecular force present in H2S(g).
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following characteristics indicates the presence of weak intermolecular forces in a liquid?
A) a low heat of vaporization
B) a high critical temperature
C) a low vapor pressure
D) a high boiling point
E) None of the above.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is not an endothermic process?
A) melting of a solid
B) vaporization
C) raising the temperature of a gas
D) condensation of water vapor
E) sublimation of dry ice
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
Indicate all the types of intermolecular forces of attraction in CH2O(g).
Correct Answer

verified
dipole-dip...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which of the responses includes all of the following that can form hydrogen bonds with water molecules? (1) Na+ (2) CH3COOH (3) C2H6 (4) CH3NH2
A) (1) and (2)
B) (1) and (3)
C) (2) and (3)
D) (2) and (4)
E) (3) and (4)
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The normal boiling point of methanol (CH3OH) is 64.6°C. Given that the vapor pressure of methanol is 75.0 torr at 15.2°C, calculate the molar enthalpy of vaporization of methanol.
A) 0.383 kJ/mol
B) 3.00 kJ/mol
C) 38.0 kJ/mol
D) 27.5 kJ/mol
E) 74.7 kJ/mol
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following substances will have both dispersion forces and dipole-dipole forces?
A) HCl
B) BCl3
C) Br2
D) H2
E) CO2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
The shape of the water-to-glass meniscus results from the strong adhesive forces between glass and water.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Choose the response that lists the member of each of the following pairs that has the higher boiling point. (I) H2O or KI (II) HF or HI (III) Cl2 or Br2
A) H2O, HF, and Cl2
B) KI, HF, and Br2
C) KI, HI, and Br2
D) H2O, HI, and Cl2
E) KI, HF, and Cl2
G) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
Of the pair of compounds given, which would have the stronger intermolecular forces of attraction? SF4 or C10H22
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Solid iodine has a vapor pressure of 1.0 mmHg at 39°C. How many moles of iodine will sublime into a 500. mL flask at this temperature? If the volume of the flask is doubled at constant temperature, what will happen to the equilibrium vapor pressure of I2? (Assume some solid I2 is always present in the container.)
A) 2.1 * 10-4 mol; vapor pressure increases
B) 2.0 * 10-2 mol; vapor pressure increases
C) 2.6 * 10-5 mol; no change in vapor pressure
D) 2.1 * 10-4 mol; no change in vapor pressure
E) 2.6* 10-5 mol; vapor pressure decreases
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Which liquid is expected to have the larger surface tension at a given temperature, CCl4 or H2O? Briefly explain.
Correct Answer

verified
Water would have the...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Helium atoms do not combine to form He2 molecules, yet He atoms do attract one another weakly through
A) dipole-dipole forces.
B) ion-dipole forces.
C) dispersion forces.
D) dipole-induced dipole forces.
E) hydrogen bonding.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Based on the phase diagram shown below, which is more dense: the liquid phase or the solid phase? 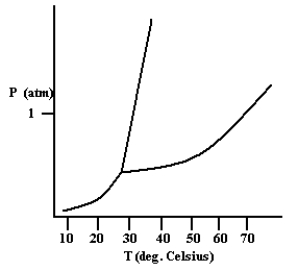
Correct Answer

verified
the solid ...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
Given the following compound and its boiling point, identify whether it is polar or nonpolar: H2S, -60.7°C.
Correct Answer

verified
Correct Answer
verified
Showing 101 - 120 of 138
Related Exams