A) 8.1 × 10-10 yr
B) 2.4 × 109 yr
C) 2.8 × 109 yr
D) 4.6 × 109 yr
E) 6.8 × 109 yr
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many 14C atoms are in a charcoal sample that has a decay rate of 3,500 disintegrations per min? (For 14C, t1/2 = 5,730 yr.)
A) 2.9 × 107 atoms
B) 8.0 × 10-7 atoms
C) 1.4 × 1014 atoms
D) 1.5 × 1013 atoms
E) 6.02 × 1020 atoms
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Define free radicals.
Correct Answer

verified
molecular fragments ...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
Radioactive nitrogen-13 has a half-life of 10 minutes.After 40 minutes, how much of this isotope would remain in a sample that originally contained 96 mg?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The isotope with the greatest nuclear binding energy per nucleon is
A) ![]() H
H
B) ![]() He
He
C) ![]() C
C
D) ![]() Fe
Fe
E) ![]() U
U
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine how much energy is released when thorium-230 decays according to ![Determine how much energy is released when thorium-230 decays according to <sup> </sup> .[Atomic masses: thorium-230 = 230.033127 amu; helium-4 = 4.002603 amu; radium-226 = 226.025403 amu] A) 3.98 × 10<sup>9</sup> kJ/mol B) 4.60 × 10<sup>8</sup> kJ/mol C) 7.20 × 10<sup>11</sup> kJ/mol D) 4.90 × 10<sup>9</sup> kJ/mol E) 7.15 × 10<sup>11</sup> kJ/mol](https://d2lvgg3v3hfg70.cloudfront.net/TB3246/11ea7cbf_8f3e_3dd0_a2ab_c563d2a78863_TB3246_11.jpg) .[Atomic masses: thorium-230 = 230.033127 amu; helium-4 = 4.002603 amu; radium-226 = 226.025403 amu]
.[Atomic masses: thorium-230 = 230.033127 amu; helium-4 = 4.002603 amu; radium-226 = 226.025403 amu]
A) 3.98 × 109 kJ/mol
B) 4.60 × 108 kJ/mol
C) 7.20 × 1011 kJ/mol
D) 4.90 × 109 kJ/mol
E) 7.15 × 1011 kJ/mol
G) None of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
When a 87Br nucleus emits a beta particle, the nuclear species that results is ___________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Cobalt-60 is a beta emitter with a half-life of 5.3 years.Approximately what fraction of the cobalt-60 atoms in a particular sample will remain after 32 years?
A) 1/6
B) 1/8
C) 1/16
D) 1/32
E) 1/64
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The following nuclear equation is correctly balanced. 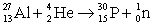
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
What type of nuclear process is illustrated below? 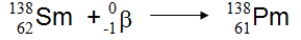
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is an example of a free radical?
A) H3O+
B) H2O2
C) HO2-
D) HO2·
E) H2O
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Complete and balance the nuclear equation 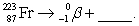
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the other product of the following nuclear transformation. 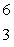 Li +
Li + 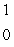 n
n 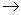 ____ +
____ + 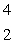 He
He
A) ![]() He
He
B) ![]() H
H
C) ![]() H
H
D) ![]()
E) ![]() H
H
G) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 121 - 133 of 133
Related Exams