A) 28.5 mol
B) 1.04 mol
C) 0.139 mol
D) 0.0285 mol
E) 0.00721 mol
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
A combination reaction may also be a displacement reaction.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In a redox reaction, electrons are transferred
A) from the substance being oxidized to the reducing agent.
B) from the oxidizing agent to the reducing agent.
C) from the substance being reduced to the oxidizing agent.
D) from the substance being oxidized to the substance being reduced.
E) from the substance being reduced to the substance being oxidized.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Covalent compounds, dissolved in water, never produce conducting solutions.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Oxidation is associated with an increase in oxidation number.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Aqueous potassium iodate (KIO3) and potassium iodide (KI) react in the presence of dilute hydrochloric acid, as shown below. KIO3(aq) + 5KI(aq) + 6HCl(aq) 3I2(aq) + 6KCl(aq) + 3H2O(l) What mass of iodine (I2) is formed when 50.0 mL of 0.020 M KIO3 solution reacts with an excess of KI and HCl?
A) 0.13 g I2
B) 0.25 g I2
C) 0.38 g I2
D) 0.76 g I2
E) none of the above
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Aqueous potassium iodate (KIO3) and potassium iodide (KI) react in the presence of dilute hydrochloric acid (HCl) , as shown below. KIO3(aq) + 5KI(aq) + 6HCl(aq) 3I2(aq) + 6KCl(aq) + 3H2O(l) What mass of iodine (I2) is formed when 15.0 mL of 0.0050 M KIO3 solution reacts with 30.0 mL of 0.010 M KI solution in the presence of excess HCl?
A) 0.023 g I2
B) 0.029 g I2
C) 0.057 g I2
D) 0.046 g I2
E) 0.076 g I2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the molarity of a 23.55-mL solution which contains 28.24 mg of sodium sulfate (used in dyeing and printing textiles, 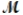 = 139.04 g/mol) .
= 139.04 g/mol) .
A) 8.625 M
B) 1.199 M
C) 0.8339 M
D) 0.2031 M
E) 0.008625 M
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
A 0.1873 g sample of a pure, solid acid, H2X was dissolved in water and titrated with 0.1052 M NaOH solution. The balanced equation for the neutralization reaction occurring is H2X(aq) + 2NaOH(aq) Na2X(aq) + 2H2O(l) If the molar mass of H2X is 85.00 g/mol, calculate the volume of NaOH solution needed in the titration.
Correct Answer

verified
Correct Answer
verified
True/False
Chemical reactions generally reach equilibrium because one of the reactants is used up.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is not a redox reaction?
A) 2H2(g) + O2(g) 2H2O(l)
B) Zn(s) + H2SO4(aq) ZnSO4(aq) + H2(g)
C) H2O(l) + NH3(g) NH4+(aq) + OH-(aq)
D) 6FeSO4(aq) + K2Cr2O7(aq) + 7H2SO4(aq) Cr2(SO4) 3(aq) + 3Fe2(SO4) 3(aq) + K2SO4(aq) + 7H2O(l)
E) Cl2(g) + 2KBr(aq) Br2(l) + 2KCl(aq)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a strong base?
A) NH3
B) Ca(OH) 2
C) Al(OH) 3
D) B(OH) 3
E) CH3OH
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A standard solution of 0.243 M NaOH was used to determine the concentration of a hydrochloric acid solution. If 46.33 mL of NaOH is needed to neutralize 10.00 mL of the acid, what is the molar concentration of the acid?
A) 0.0524 M
B) 0.888 M
C) 1.13 M
D) 2.26 M
E) 2.43 M
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the classification for the following reaction. Fe(s) + 2Fe3+(aq) 3Fe2+(aq)
A) precipitation
B) acid-base
C) redox
D) decomposition
E) none of the above
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calcium chloride is used to melt ice and snow on roads and sidewalks and to remove water from organic liquids. Calculate the molarity of a solution prepared by diluting 165 mL of 0.688 M calcium chloride to 925.0 mL.
A) 3.86 M
B) 0.743 M
C) 0.222 M
D) 0.123 M
E) 0.114 M
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the oxidation number of the chlorine in perchloric acid, HClO4, a strong oxidizing agent.
A) -1
B) +4
C) +5
D) +7
E) None of the above is the correct oxidation number.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles of H+(aq) ions are present in 750 mL of 0.65 M hydrochloric acid?
A) 1.2 mol
B) 0.98 mol
C) 0.87 mol
D) 0.65 mol
E) 0.49 mol
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following ionic compounds is soluble in water?
A) Na2S
B) PbI2
C) AgCl
D) CuS
E) Ca3(PO4) 2
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Copper(II) sulfide, CuS, is used in the development of aniline black dye in textile printing. What is the maximum mass of CuS which can be formed when 38.0 mL of 0.500 M CuCl2 are mixed with 42.0 mL of 0.600 M (NH4) 2S? Aqueous ammonium chloride is the other product.
A) 2.41 g
B) 1.82 g
C) 1.21 g
D) 0.909 g
E) 0.044 g
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many mL of concentrated nitric acid (HNO3, 16.0 M) should be diluted with water in order to make 2.00 L of 2.00 M solution?
A) 32.0 mL
B) 62.5 mL
C) 125 mL
D) 250. mL
E) 500. mL
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 124
Related Exams