A) the definition of absolute zero.
B) the definition of an ideal gas.
C) the conservation of energy.
D) thermal expansion.
E) the conservation of momentum.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
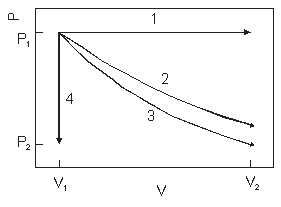 The diagram above show the state of an ideal gas going from (V1,P1) to a final state. Which path best represents adiabatic expansion?
The diagram above show the state of an ideal gas going from (V1,P1) to a final state. Which path best represents adiabatic expansion?
A) 1
B) 2
C) 3
D) 4
E) none of the paths
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For most metals, the heat capacity goes as C = T + AT3 at low temperatures (~ 100 K) , whereas insulators only has the T3 term. Both and A are constants. The two terms in the expression for C suggests
A) there are at least two degrees of freedom in metals.
B) there are two independent mechanisms for heat to be absorbed or released in metals.
C) that there are two kinds of heat that are absorbed by metals.
D) that thermal energy is transferred to metals differently than insulators.
E) nothing. It just happens that way.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the following to answer question : 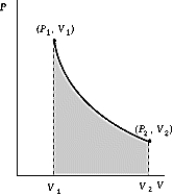 -An ideal gas initially at 50ºC and pressure P1 = 100 kPa occupies a volume V1 = 3 L. It undergoes a quasistatic, isothermal expansion until its pressure is reduced to 50 kPa. How much heat enters the gas during this process? R = 8.314 J/mol·K = 8.206 L·atm/mol·K.
-An ideal gas initially at 50ºC and pressure P1 = 100 kPa occupies a volume V1 = 3 L. It undergoes a quasistatic, isothermal expansion until its pressure is reduced to 50 kPa. How much heat enters the gas during this process? R = 8.314 J/mol·K = 8.206 L·atm/mol·K.
A) 116 J
B) 208 J
C) 256 J
D) 304 J
E) 416 J
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the following to answer question : 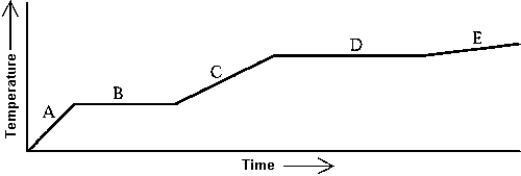 -Heat is added to a substance at a constant rate. The substance starts as a solid and is melted; the liquid is heated and vaporized; finally, the vapor is heated. This process is shown in the graph. The specific heat of the solid can be found by
-Heat is added to a substance at a constant rate. The substance starts as a solid and is melted; the liquid is heated and vaporized; finally, the vapor is heated. This process is shown in the graph. The specific heat of the solid can be found by
A) multiplying the length of B (in seconds) by the rate at which heat is added, and dividing by the mass of the substance.
B) multiplying the length of D (in seconds) by the rate at which heat is added, and dividing by the mass of the substance.
C) dividing the rate at which heat is added by the product of the slope of A and the mass of the substance.
D) dividing the rate at which heat is added by the product of the slope of C and the mass of the substance.
E) dividing the rate at which heat is added by the product of the slope of E and the mass of the substance.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A system has a heat capacity of 100 J. This means
A) it is possible to extract the 100 J of heat and convert it to work.
B) it is possible to transfer the 100 J of heat to the environment.
C) some of the heat capacity can be converted to work.
D) some of the heat capacity can be transferred to another system if there is a temperature difference.
E) (C) and (D)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If the heat capacities of both ice and steam are 0.5 cal/g · Cº, the quantity of heat required to change 1 g of ice at -10ºC to steam at 120ºC is approximately
A) 750 cal
B) 735 cal
C) 630 cal
D) 620 cal
E) 555 cal
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In a certain thermodynamic process, 1000 cal of heat are added to a gas confined in a cylinder. At the same time, 1000 J of work are done by the gas as it expands. The increase in internal energy of the gas is
A) zero
B) 3186 J
C) -239 J
D) 5186 J
E) 1239 J
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An ideal monatomic gas has a molar heat capacity Cmp at constant pressure. What is the molar heat capacity at constant volume of an ideal diatomic gas?
A) Cmp
B) Cmp + R
C) Cmp - R
D) Cmp + 3R/2
E) Cmp - 3R/2
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 1.0-kg piece of marble at 100ºC is dropped into 2.5 kg of water at 1.0ºC and the resulting temperature is 7.0ºC. The specific heat of the marble is approximately
A) 0.16 kcal/kg · Cº
B) 0.75 kcal/kg · Cº
C) 0.61 kcal/kg · Cº
D) 0.30 kcal/kg · Cº
E) 0.26 kcal/kg · Cº
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the following to answer question : 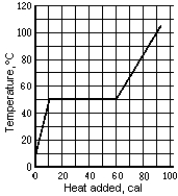 -The graph shows the temperature of a 1.0-g sample of material as heat is added to it. The material is initially a solid at 10ºC. The pressure remains constant, and there is no chemical change. The heat of fusion of the material is
-The graph shows the temperature of a 1.0-g sample of material as heat is added to it. The material is initially a solid at 10ºC. The pressure remains constant, and there is no chemical change. The heat of fusion of the material is
A) 10 cal/g
B) 50 cal/g
C) 30 cal/g
D) 90 cal/g
E) None of these is correct.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
During a certain thermodynamic process, 418 J of work are done on a system and 214 cal of heat are transferred to the system. The change in internal energy during the process is
A) 314 cal
B) 114 cal
C) 468 cal
D) 368 cal
E) 632 cal
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the following to answer question :  -Heat is added to a substance at a constant rate. The substance starts as a solid and is melted; the liquid is heated and vaporized; finally, the vapor is heated at constant volume. This process is shown in the graph. The specific heat at constant volume of the vapor can be found by
-Heat is added to a substance at a constant rate. The substance starts as a solid and is melted; the liquid is heated and vaporized; finally, the vapor is heated at constant volume. This process is shown in the graph. The specific heat at constant volume of the vapor can be found by
A) multiplying the length of B (in seconds) by the rate at which heat is added, and dividing by the mass of the substance.
B) multiplying the length of D (in seconds) by the rate at which heat is added, and dividing by the mass of the substance.
C) dividing the rate at which heat is added by the product of the slope of A and the mass of the substance.
D) dividing the rate at which heat is added by the product of the slope of C and the mass of the substance.
E) dividing the rate at which heat is added by the product of the slope of E and the mass of the substance.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A container contains a 200 mL of 100% proof alcohol (i.e., it has 50% ethyl alcohol and 50% water by volume) at 20 C. How much heat is needed to bring the mixture to the boiling point of the alcohol? (assume that the specific heat in the 100% proof can be treated as due to the alcohol and water separately, and the density, boiling point and specific heat of alcohol are 0.81 g/cm3, 78 C, and 2.4 J/(g.C ) , respectively)
A) 32650 J
B) 35525 J
C) 11136 J
D) 17400 J
E) 48557 J
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
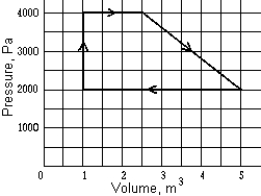 A reversible heat engine has the PV graph shown. The net work performed in one cycle is approximately
A reversible heat engine has the PV graph shown. The net work performed in one cycle is approximately
A) zero
B) 2.0 kJ
C) 4.2 kJ
D) 5.5 kJ
E) 10 kJ
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the following to answer question :  -One mole of an ideal gas ( = 5/3) expands adiabatically and quasistatically from a pressure P1 = 3 atm and a temperature of 30ºC to a pressure P2 = 1 atm. How much work is done by the gas during this process? R = 8.314 J/mol·K = 8.206 L·atm/mol·K.
-One mole of an ideal gas ( = 5/3) expands adiabatically and quasistatically from a pressure P1 = 3 atm and a temperature of 30ºC to a pressure P2 = 1 atm. How much work is done by the gas during this process? R = 8.314 J/mol·K = 8.206 L·atm/mol·K.
A) 50.3 kJ
B) 63.5 kJ
C) 95.9 kJ
D) 131 kJ
E) 158 kJ
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
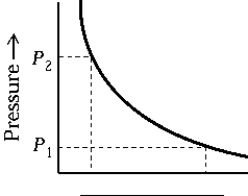 The work done by a gas on a piston can be obtained from the graph, provided the abscissa represents the
The work done by a gas on a piston can be obtained from the graph, provided the abscissa represents the
A) internal energy.
B) temperature.
C) density.
D) volume.
E) time.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
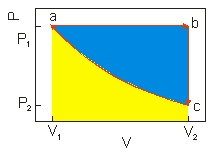 An ideal gas undergoes a cyclic expansion and compression along the path a b c a as shown above. The work done along c a of the cycle is the area
An ideal gas undergoes a cyclic expansion and compression along the path a b c a as shown above. The work done along c a of the cycle is the area
A) shaded blue.
B) shaded yellow.
C) shaded blue and yellow.
D) negative of the area shaded blue.
E) negative of the area shaded yellow.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the following to answer question : 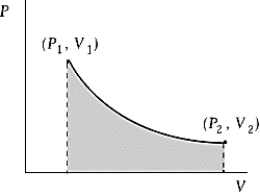 -An ideal gas initially at 100ºC and pressure P1 = 250 kPa occupies a volume V1 = 4.5 L. It undergoes a quasistatic, isothermal expansion until its pressure is reduced to 150 kPa. How much does the internal energy of the gas change during this process? R = 8.314 J/mol·K = 8.206 L·atm/mol·K.
-An ideal gas initially at 100ºC and pressure P1 = 250 kPa occupies a volume V1 = 4.5 L. It undergoes a quasistatic, isothermal expansion until its pressure is reduced to 150 kPa. How much does the internal energy of the gas change during this process? R = 8.314 J/mol·K = 8.206 L·atm/mol·K.
A) 116 J
B) 320 J
C) 575 J
D) 640 J
E) The internal energy does not change during this process.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The specific heat of a substance is a measure of
A) energy needed to change the kinetic energies of atoms and/or molecules and/or electrons by a temperature increment.
B) total energy in the substance for a given temperature change.
C) energy needed to vaporize/liquefy the substance.
D) energy needed to liquefy/solidify the substance.
E) none of the above
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 114
Related Exams